Michael J. Hawken
Cortical circuits and neuronal mechanisms of visual perception
My main research interest is defining the role of cortical neurons in visual processing.
This work includes physiological and anatomical studies in the primate visual pathways, mainly
the primary visual cortex. In conjunction with these physiological studies, I collaborate in
studies that relate pursuit eye movements to perceptual performance.
Cortical Processing and Circuits: Much of my visual physiology work has centered on unraveling
the cortical receptive field properties in adult monkey cortex. This is important for a number
of reasons: visual cortex is one of the best understood cortical regions and appears to play a
pivotal role in many of the features of visual analysis underlying visual perception. It is
also strongly affected by deprivation and learning so having a solid understanding of the
normal properties is essential to understanding dysfunction. My colleague, Andrew Parker, and
I showed that the activity of individual neurons could match perceptual performance
(Parker & Hawken, 1985; Barlow et al, 1987; Hawken & Parker, 1990) on a number of low-level
visual tasks. With Andrew Parker while at Oxford and with my colleague Bob Shapley since I
moved to NYU I have also studied how receptive fields are organized spatially (Hawken & Parker,
1987; Parker & Hawken, 1988; Hawken & Parker, 1991), and how different streams from the retina,
the M and P-pathways, selectively influence the properties of different streams of cortical
neurons (Hawken & Parker, 1984; Hawken et al, 1988),
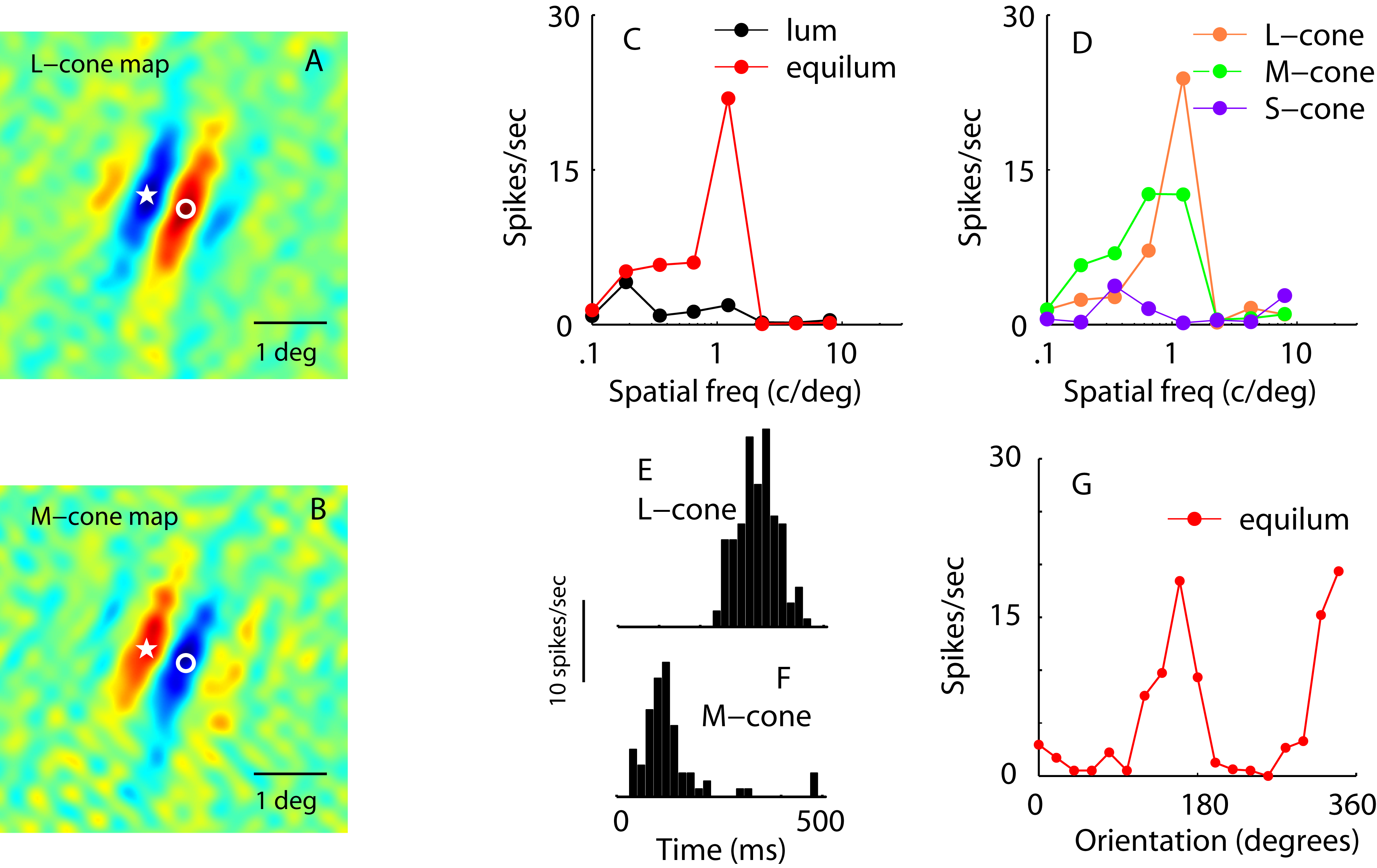 the temporal selectivity of cortical
neurons (Hawken et al, 1996), the influence of surround suppression on receptive fields
(Sceniak et al, 1999; 2001; 2002), organization of orientation selectivity (Ringach et al,
2002) and the chromatic organization of receptive fields (Johnson et al, 2001; 2004).
From these latter studies we proposed a new view of chromatically double opponent receptive
fields (Shapley & Hawken, 2002; Shapley & Hawken, 2011). Overall we have begun to build up a comprehensive picture
of the complexity of receptive field organization in V1 and have developed a large database
that we believe will offer important insights into cortical function.
the temporal selectivity of cortical
neurons (Hawken et al, 1996), the influence of surround suppression on receptive fields
(Sceniak et al, 1999; 2001; 2002), organization of orientation selectivity (Ringach et al,
2002) and the chromatic organization of receptive fields (Johnson et al, 2001; 2004).
From these latter studies we proposed a new view of chromatically double opponent receptive
fields (Shapley & Hawken, 2002; Shapley & Hawken, 2011). Overall we have begun to build up a comprehensive picture
of the complexity of receptive field organization in V1 and have developed a large database
that we believe will offer important insights into cortical function.
In a related and complementary series of studies with Bob Shapley we have used techniques of
reverse correlation that were developed by Dario Ringach when he was a student in our lab to
investigate the mechanisms underlying receptive field organization (Ringach et al, 1997). From
these studies (Ringach et al, 2003; Xing et al, 2004; 2005; 2006) we have proposed how excitatory
and suppressive mechanisms are involved in the generation of orientation selectivity (Shapley
et al, 2003; Xing et al, 2011) and spatial frequency tuning (Ringach et al, 2002; Xing et al, 2004).
Structure-Function Relationships in Primary Visual Cortex: On the one hand we often
have detailed knowledge of neuronal firing to an array of visual stimuli, obtained from
extracellular recording, that characterize the receptive field.
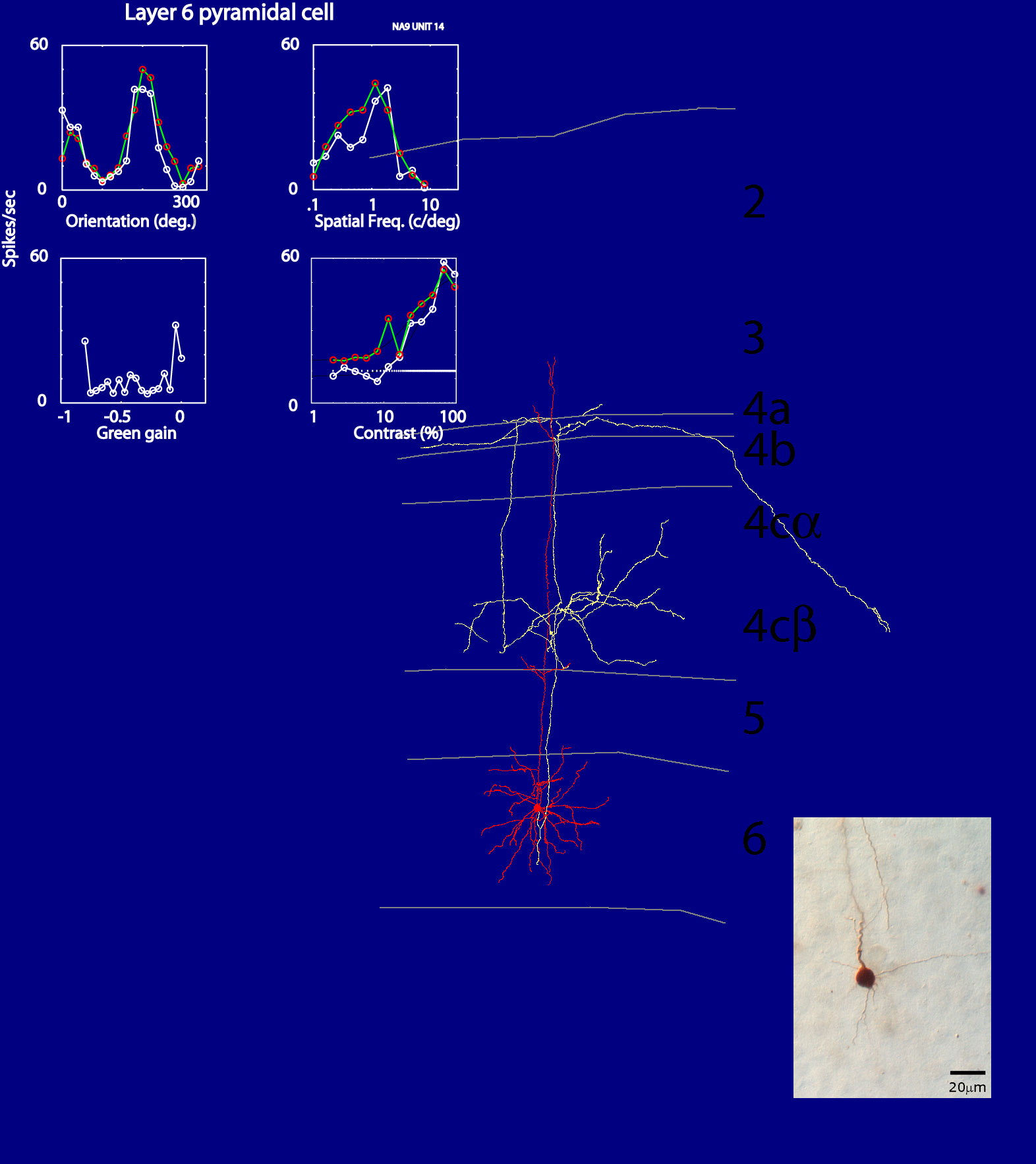 On the other hand, from anatomical studies, we know about the intricate neuronal architecture
and the patterns of synaptic connections both within V1 and between V1 and extra-striate cortex.
In order to better understand such a complicated neuronal network, modeling how cortical neurons
interact within the network is a crucial area of research. At present, there is one link that is
missing that if found would enable much more realistic modeling of the cortex: the link between structure
and function. In collaboration with Siddhartha Joshi I have recently developed a method called
juxtacellular loose-patch (JC-LP) recording (Joshi & Hawken, 2006) to label neurons in
primary visual cortex after functionally characterizing their receptive field properties. One
of out goals is to identify subclasses of excitatory and inhibitory neurons and associate them
with receptive field properties, thereby greatly extending our current knowledge of how the
cortical circuits operate.
On the other hand, from anatomical studies, we know about the intricate neuronal architecture
and the patterns of synaptic connections both within V1 and between V1 and extra-striate cortex.
In order to better understand such a complicated neuronal network, modeling how cortical neurons
interact within the network is a crucial area of research. At present, there is one link that is
missing that if found would enable much more realistic modeling of the cortex: the link between structure
and function. In collaboration with Siddhartha Joshi I have recently developed a method called
juxtacellular loose-patch (JC-LP) recording (Joshi & Hawken, 2006) to label neurons in
primary visual cortex after functionally characterizing their receptive field properties. One
of out goals is to identify subclasses of excitatory and inhibitory neurons and associate them
with receptive field properties, thereby greatly extending our current knowledge of how the
cortical circuits operate.
Modulation and Inhibition in Cortical Circuits: Primarily driven by recent work with a
predoc and then postdoc in my lab, Anita Disney, I have begun to study how neurons in primary
visual cortex are influenced by neuromodulators. It has been known for sometime that that there
is an extensive innervation of cortex by cholinergic afferents, and the effects of ACh
application to cortical neurons have shown mixed effects. Anita showed in her thesis work that
one class of receptors for acetylcholine (ACh), the nicotinic receptors, are located on primary
afferents arriving from the geniculate into layer 4c of monkey primary visual cortex. She
hypothesized that nicotine should enhance geniculate transmission, and we devised a series of
experiments to investigate this in vivo. We found that the response of neurons in layer 4c
was enhanced by the iontophoretic application of nicotine, but neurons outside 4c were either
unaffected or suppressed. Tantalizingly, the effects we observed are almost identical to those
that are observed during an enhancement of response during attention in the awake-monkey,
which prompted us to suggest that some of the effects of attention might be mediated by ACh
via nicotinic receptors (Disney et al, 2007).
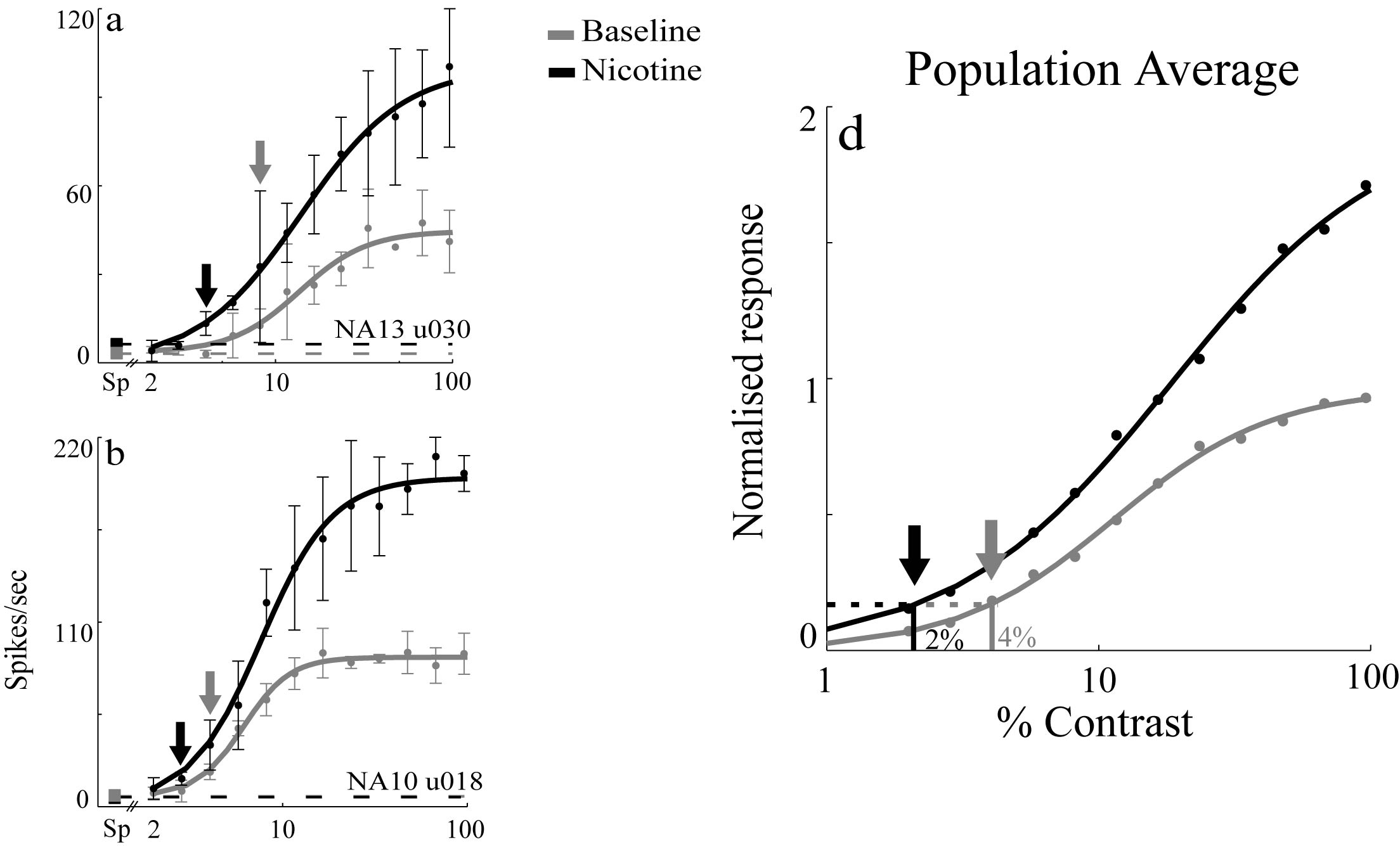
Most of the receptors for ACh on inhibitory interneurons in primate cortex are muscurinic. We
found that outside layer 4c most effects of ACh are suppressive. We also found that blockers of the inhibitory
receptor, GABA, block the suppressive effects of ACh in cortex. We have proposed a model that
includes both facilitation via ACh on presynpatic nAChR's on LGN afferent terminals in cortex
and broadly tuned ACh induced suppression via mAChR's on inhibitory interneurons in cortex
(Disney et al, 2012). This model produces selective response enhancement without loss of tuning.
In addition, we have recently determined the distribution of neurons in primate V1
with the Kv3.1 and Kv3.2 ion channels that confer the fast-spiking phenotype on neurons,
finding that in addition to the expected localization on sub-populations of inhibitory
interneurons there is also expression on excitatory neurons (Constantinople et al, 2009).
These results suggest that, unlike rodent cortex, primate cortex is likely to have a population
of fast-spiking excitatory neurons.
Color and Motion Psychophysics and Eye Movements: In the 1980� s it was speculated that there
were selective mechanisms for the perception of color and motion, and that these had their own
separate pathways in the brain. In a series of psychophysical studies with Karl Gegenfurtner,
started when he was a postdoc here at NYU and continued since he moved back to Germany, we
found that stimuli that selectively isolate the chromatic pathway can provide signals to drive
the perception of motion (Hawken et al, 1994; Gegenfurtner & Hawken, 1995; 1996; Hawken &
Gegenfurtner, 1999).
Perception and action are intimately related. One intriguing question that has received a lot
of attention recently is whether the same neural signals that are used to perceive visual
objects are also used to initiate and maintain actions. In a study that formed his Ph.D. thesis
I worked with Dario Ringach on recording vergence eye movements in response to a stimulus
where the three-dimensional perception of depth comes from motion cues, but is not due to
disparity (Ringach et al, 1996). In this study we found that the percept of motion in depth
was strongly related to the vergence eye movements, suggesting that the same processing is used
for perception as for driving motor action.
Another eye movement system that is particularly amenable to measurement is the pursuit system,
it is voluntary and can be studied in the open loop phase. In a series of studies we sought to
determine whether the motion signals that drive perception are the same as those that drive
pursuit (Hawken & Gegenfurtner, 2001; Gegenfurtner et al, 2003; Spering et al, 2005; Braun
et al, 2008) and, surprisingly, found that although they are strongly correlated they are
not the same. These results suggest that, under some conditions, signals driving perception
and action are distinct.
E-mail: mjh2@nyu.edu
Representative Publications
Ringach, D.L., Hawken, M.J. & Shapley R.M. (2002) Receptive field structure of neurons in monkey
primary visual cortex revealed by stimulation with natural image sequences. Journal of Vision, 2,
12-24.
pdf
Ringach, D.L., Bredfeldt, C.E., Shapley, R.M. & Hawken, M.J. (2002) Suppression of
neural responses to non-optimal stimuli enhances tuning selectivity in macaque V1.
Journal of Neurophysiology, 87, 1018-1027.
pdf
Ringach, D.L., Shapley, R.M. & Hawken, M.J. (2002) Diversity and laminar
specialization of orientation selectivity in macaque V1. Journal of Neuroscience, 22,
5639-5651.
pdf
Shapley, R. & Hawken, M.J. (2002) Neural mechanisms for color perception in the
primary visual cortex. Current Opinion in Neurobiology, 12, 426-432.
pdf
Sceniak, M.P., Hawken, M.J. & Shapley, R.M. (2002) Contrast dependent changes in
spatial frequency tuning: effects of a changing receptive field size. Journal of
Neurophysiology, 88, 1363-1373.
pdf
Ringach, D.L., Hawken, M.J. & Shapley, R.M. (2003) Dynamics of orientation tuning in
macaque V1: the role of global and tuned suppression. Journal of Neurophysiology,
90, 342-352.
pdf
Shapley, R.M., Hawken, M.J. & Ringach, D.L. (2003) Dynamics of orientation tuning in
primary visual cortex and the importance of cortical inhibition. Neuron 38, 689-699.
pdf
Kiorpes, L., Tang, C., Hawken, M.J. & Movshon, J.A. (2003) Ideal observer analysis of
the development of spatial contrast sensitivity in macaque monkeys. Journal of Vision
3, 630-641.
pdf
Gegenfurtner, K.R., Scott, B., Xing, D. & Hawken, M.J. (2003) A comparison of pursuit
eye movements and perecptual performance in speed discrimination. Journal of
Vision 3, 665-876.
pdf
Johnson, E.N., Hawken, M.J. & Shapley, R.M. (2004) Cone inputs in macaque primary
visual cortex. Journal of Neurophysiology 91, 2501-2514.
pdf
Xing, D., Ringach, D.L., Shapley R.M. & Hawken M.J. (2004) Correlation of local and
global orientation and spatial frequency tuning in macaque V1. Journal of Physiology
557, 923-933.
pdf
Williams, P.E., Mechler, F., Gordon, J., Shapley, R.M. & Hawken, M.J. (2004)
Entrainment to video displays in primary visual cortex of macaque and humans.
Journal of Neuroscience 24: 8278-8288.
pdf
Movshon, J.A., Kiorpes, L., Hawken, M.J. & Cavanaugh, J.R. (2005) Maturation of the
macaque's lateral geniculate neurons. Journal of Neuroscience 25: 2112-2122.
pdf
Xing, D., Shapley R.M., Hawken M.J. & Ringach, D.L. (2005) The effect of stimulus
size on the dynamics of orientation selectivity in Macaque V1. Journal of
Neurophysiology 94: 799-812.
pdf
Spering, M., Kerzel, D., Braun, D.I., Hawken, M.J. & Gegenfurtner, K.R. (2005) Effects
of contrast on smooth pursuit eye movements. Journal of Vision 5: 455-465.
pdf
Joshi, S. & Hawken M.J. (2006) Loose-patch-juxtacellular recording in vivo-A method
for functional characterization and labeling of neurons in macaque V1.
Journal of Neuroscience Methods, 156, 37-49.
pdf
Shapley, R.M, Hawken, M.J. & Xing, D. (2007) The dynamics of visual responses in
primary visual cortex. Progress in Brain Research 165, 21-32.
pdf
Disney, A.A., Aoki, C. & Hawken, M.J. (2007) Nicotinic receptors on thalamic
afferents enhance visual responsiveness in macaque V1. Neuron 56, 701 - 713.
pdf
pdf_suppl
Johnson, E.B., Hawken, M.J. & Shapley R.M. (2008) Color and orientation selectivity
in macaque V1. Journal of Neuroscience 28, 8096-8106.
pdf
Braun, D.I., Mennie, N., Rasche, C., Hawken, M.J. & Gegenfurtner, K.R. (2008) Smooth
pursuit eye movements to isoluminant targets. Journal of Neurophysiology 100, 1287-1300.
pdf
Constantinople, C., Disney, A.A., Maffie, J., Rudy, B. & Hawken, M.J. (2009) A
quantitative analysis of neurons with Kv3 potassium channel subunits Kv3.1b
and Kv3.2 in macaque primary visual cortex. Journal of Comparative Neurology,
516, 291-311.
pdf
Shapley R.M & Hawken, M.J. (2011) Color in the cortex: single and double-opponent
cells. Vision Research 51, 701-717.
Xing, D., Ringach, D.L., Hawken, M.J. & Shapley R.M (2011) The contribution of untuned
suppression to orientation selectivity in macaque V1. Journal of Neuroscience 31, 15972-15982.
Lee, B.B., Shapley, R.M., Hawken, M.J. & Sun, H. (2012) Spatial distribution of
cone inputs to cells of the parvocellular pathway investigated with cone-isolating
gratings. Journal of the Optical Society of America A 29, 223-232.
Garcia-Marin, V., Ahmed, T.A., Azfal, Y. & Hawken, M. J. (2012) The distribution
vesicular glutamate transporter (VGluT2) in the primary visual cortex of macaque
and human. Journal of Comparative Neurology DOI: 10.1002/cne.23165
Disney, A.A., Aoki, C.A. & Hawken, M.J. (2012) Cholinergic suppression of visual
responses in primate V1 is mediated by GABAergic inhibition. Journal of Neurophysiology
108, 1907-1923.
 the temporal selectivity of cortical
neurons (Hawken et al, 1996), the influence of surround suppression on receptive fields
(Sceniak et al, 1999; 2001; 2002), organization of orientation selectivity (Ringach et al,
2002) and the chromatic organization of receptive fields (Johnson et al, 2001; 2004).
From these latter studies we proposed a new view of chromatically double opponent receptive
fields (Shapley & Hawken, 2002; Shapley & Hawken, 2011). Overall we have begun to build up a comprehensive picture
of the complexity of receptive field organization in V1 and have developed a large database
that we believe will offer important insights into cortical function.
the temporal selectivity of cortical
neurons (Hawken et al, 1996), the influence of surround suppression on receptive fields
(Sceniak et al, 1999; 2001; 2002), organization of orientation selectivity (Ringach et al,
2002) and the chromatic organization of receptive fields (Johnson et al, 2001; 2004).
From these latter studies we proposed a new view of chromatically double opponent receptive
fields (Shapley & Hawken, 2002; Shapley & Hawken, 2011). Overall we have begun to build up a comprehensive picture
of the complexity of receptive field organization in V1 and have developed a large database
that we believe will offer important insights into cortical function.

